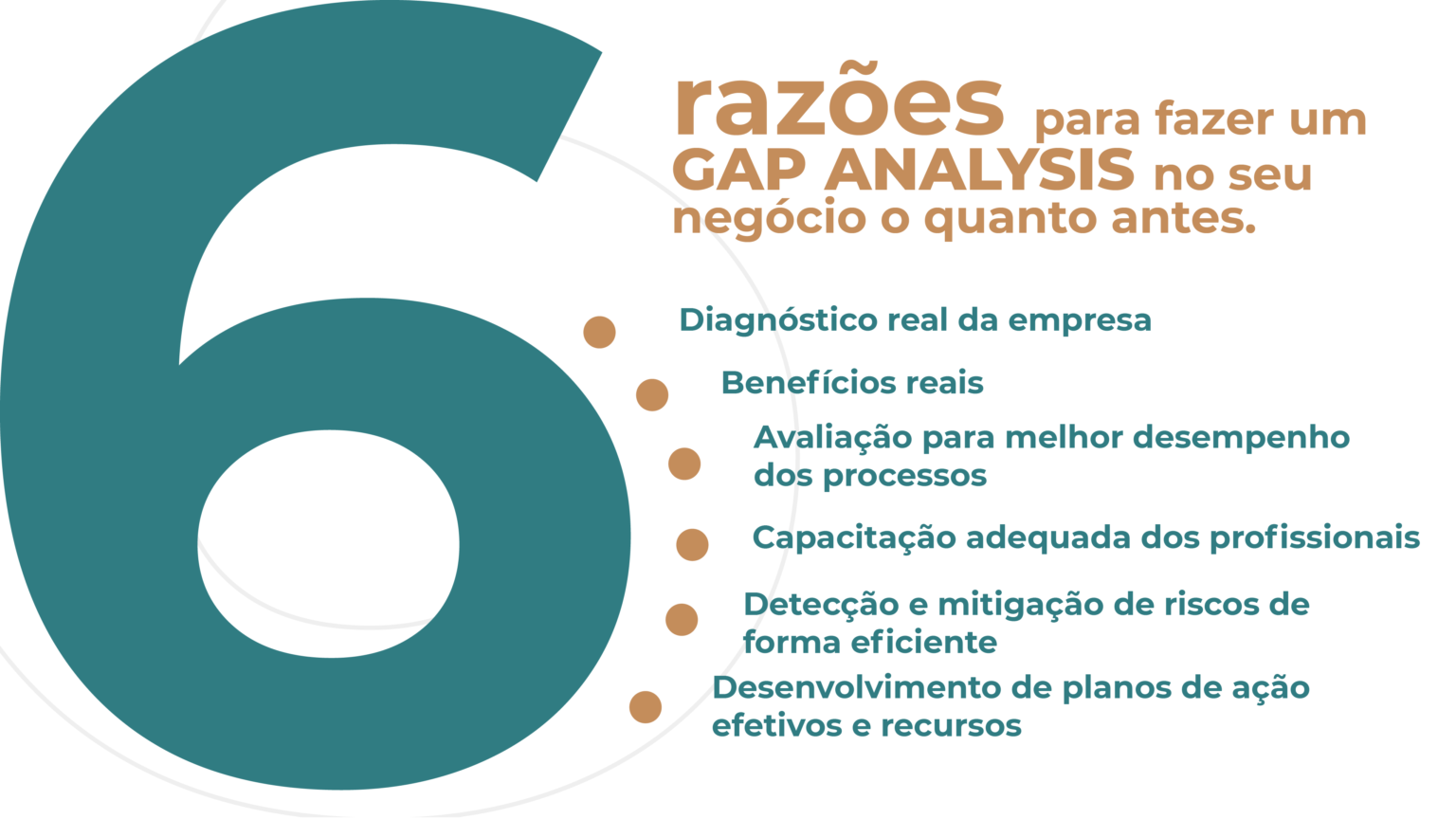
On a day-to-day high, companies are in the process of numerous operational processes. In addition to the activities that take place within the departments themselves, the big challenge is the integration of activities between the areas of the company, team engagement and the gain of agility that the business requires to remain competitive in the market.
The fact is that in the midst of this whole scenario, there may be failures that require to be identified and resolved immediately in order not to impact the performance of the organization and, especially, ensure the quality of the service or product being delivered to the customer.
This is where the Quality Management System (QMS) plays a key role, contributing to the appointment and rapid management of fault corrections (non-conformities, what we call “risk management”) in corporate processes.
How to measure the risks of QMS?
The definition of risk measurement metrics, to be considered in the company’s Quality Management System, will depend on the requirements and regulations of the industry that the business is inserted.
Mapping processes and recording the measurement of identified risks and opportunities for improvement is extremely important, especially with regard to audits. This is because it helps in continuous monitoring of fault resolution, certifying good practices and positive results in the short, medium and long term.
In addition, risk measurement, mapping of opportunities and monitoring of corrective actions for non-conformities become a fundamental support in the preparation and new reviews of the company’s strategic planning.
Implementing culture between teams
Among the major challenges of implementing the QMS is team engagement. In this sense, it is necessary to adopt actions that promote the autonomy and recognition of employees in relation to projects.
Some of the measures that Kivalita Consulting adopts, for example, aimed at changing the culture to promote quality in the company as a whole are:
- Training (technical and soft skills);
- Periodic follow-ups for the determination of activities, deliveries and feedbacks;
- Risk management on the company’s processes (both for new or existing processes);
There is no perfect Quality Management System for your company, but certainly continuous improvement should be implemented as a culture to teams and be part of the organization’s DNA, prioritizing respect for good practices required by your industry legislation.
Benefits of the “Continuous K” solution for continuous improvement
Have you met Kivalita Consulting? We are a Brazilian company specialized in the development of quality management and validation of technologies for companies in the area of Life Science.
Thus, our expertise is to meet the most stringent regulatory and market requirements, with competence to support organizations of various sizes of this segment, such as pharmaceutical, food, cosmetic and chemical industries, among others.
Among the solutions in our portfolio is Continuos K, which is based on the oldest quality methodology and more difficult to be maintained in companies: the PDCA (Plan-Do-Check-Act). Thus, we act in the definition of processes to avoid rework, risks and losses, sustaining the “continuous” growth of your business. For this, we offer:
- Quality advice and regulatory technology, in the face of specific demands of the sector;
- Monthly follow-up meetings;
- Monthly training;
- Attendance, reports and weekly dashboards of the project with indicators for decision making;
- Advice for adequacy to certification (ISP/ONA)
- Management and planning of activities via platform, in real time
The great differential of this solution is that itis carried out in a resume (online) way.
Having a Quality Management System is undoubtedly a powerful strategic advantage, able to position the organization at a new level in terms of quality and operational performance.
So, let’s talk about SGQ for your company? Contact Kivalita Consulting, it will be a pleasure to serve you!