Who identifies: “Instrumental analysis class on chromatography. The Professor passes a lot of theory on the subject and pours several equations, graphs, complicated terms, turns to the class and says that the professional who knows chromatography is well valued in the market, because it is a very complicated and wide-use technique.”
After class you think, “I’m never going to be valued, this is very complicated.”
I was in a class where I was presented with the chromatographic methods, and when I left, I didn’t want to see it there or painted gold in front of me. However, life plays a few pieces on us and when I came into the job market I came across the technique as one of the first great challenges I had to face, and when using in everyday life I realized that it was not such a complicated technique, but a technique that requires a lot from the user and that does not have as much complexity as the equations and graphs that were used to develop it. In this article I want to translate in a simple way the equation of Van Deemter and help those who are starting, or who already work, but do not understand well the terms and what happens during chromatography.
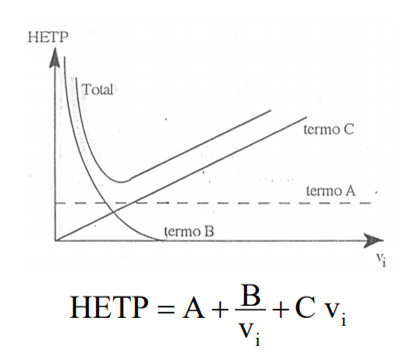
First, van deemter’sequation:
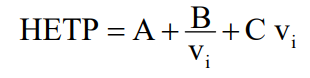
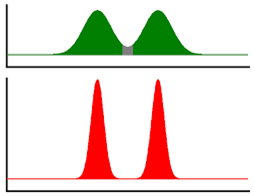
HEPT: Equivalent height of a theoretical dish – Translating: Separation efficiency, the lower this value, means that the separations will be better and the peak will tend to have a thinner profile. Figure 1: Alteration of chromatograms with the decrease in theoretical dish height.
A: Eddy diffusion term, related to column packaging. – Translating: This term is related to the quality of the column, the way particles are stored in the column, the shape of the particles and the size of the particles. To reduce the influence of this term, it is necessary to have a column with uniform particle size, uniform (spherical) formats and packaging must be standardized, and here in this last aspect we can also mention some good practices of use to increase the life of the column, such as increasing the flow gradually, not reversing the column, using suitable solvents and filtered mobile phases, all this to reduce the creation of “preferred paths” or uneven packaging.

Figure 2: Formation of preferential paths due to the non-uniformity of column particles, particles with non-uniform shapes, misusing the column,
B: Dispersion coefficient term – Translating: This term is nothing more than the displacement of the analyth during chromatographic analysis and is inversely linked to the increase in speed, that is, the higher the speed of the mobile phase the lower the diffusion of the analysis and consequently the peak will be thinner and the analysis will be faster as well (here has some limitations such as equipment pressure, column particle size… that we will not discuss at the moment.).
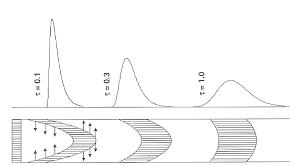
Figure 3: Influence of the speed of the mobile phase on the retention time and the shape of the peak.
C: Resistance term during mass transfer – Translating: the analyte, the moving phase and the stationary phase have interactions between them. The stationary phase will exert a resistance to the passage of the mobile phase along with the analyte and it increases with the increase of the linear velocity, or the flow of the moving phase.
V: Linear speed – Translating: Nothing less than the flow of the mobile phase.
The result of all this will result in the chart below:
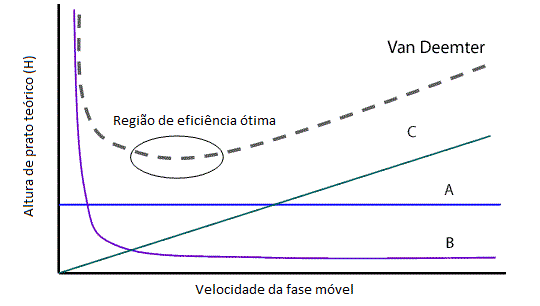
The dotted line shows the representation of van deemter’s equation, in it we can see a region where we have an ideal moving phase speed to obtain the highest separation efficiency. And how did we get to this region? Experimentally we can optimize the chromatographic conditions (composition of mobile phase, stationary phase) and change the flow to check whether the sections improve or not (in this case the improvement will occur in the profile of the chromatogram, with thinner and well resolved peaks).
Needing any help with this process on your company? Talk to us at https://www.kivalitaconsulting.com/contact
